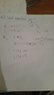Work done on ideal monatomic gas in adiabatic process
Click For Summary
Homework Help Overview
The discussion revolves around the work done on an ideal monatomic gas during an adiabatic process, focusing on the thermodynamic principles involved and the relevant equations.
Discussion Character
- Exploratory, Conceptual clarification, Assumption checking
Approaches and Questions Raised
- Participants discuss the relationship between pressure and volume in adiabatic processes, with one noting that PV is not constant. There is also mention of the work done being represented by the area under the P-V curve.
Discussion Status
The discussion is ongoing, with participants exploring different interpretations of the problem. Some guidance has been offered regarding the calculation of variables and the nature of work in thermodynamic systems.
Contextual Notes
There are indications of missing information regarding specific calculations and the need for clearer communication of questions, as some participants suggest typing out queries instead of posting images.