Discussion Overview
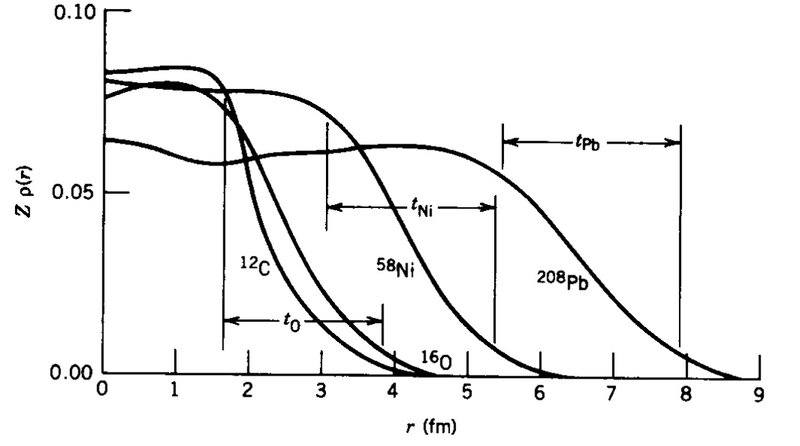
The discussion revolves around the distribution of nucleons within atomic nuclei, referencing a diagram from Krane's "Introductory Nuclear Physics." Participants explore the characteristics of nucleon distribution, particularly how it varies with mass number and the implications of the strong force on this distribution.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- Some participants assert that nucleons do not cluster around the center of the nucleus but instead have a fairly constant distribution throughout, with the number of nucleons per unit volume remaining relatively stable.
- It is noted that the distribution of nucleons tapers off, with smaller mass number nuclei exhibiting a quicker tapering compared to larger mass number nuclei, though the reasons for this behavior are not fully understood.
- One participant suggests that the tapering of nucleon distribution is related to the range of the strong force, which binds nucleons together and has a limited range.
- Another participant mentions that the charge densities of nucleons drop off slowly, indicating that there is no sharp outer boundary to the nucleus, which is supported by Krane's assertion that the distance over which charge density decreases is nearly independent of nucleus size.
- A question is raised about whether the lack of a sharp outer boundary implies that larger nuclei have a longer distance over which charge density drops, suggesting a potential relationship between nucleus size and charge density distribution.
Areas of Agreement / Disagreement
Participants generally agree on the notion that nucleons have a constant distribution and that this distribution tapers off due to the strong force. However, there is no consensus on the implications of this tapering in relation to mass number and charge density, indicating multiple competing views remain.
Contextual Notes
Some assumptions about the nature of nucleon distribution and the effects of the strong force are present but not fully explored. The discussion also reflects uncertainty regarding the relationship between nucleus size and charge density drop-off.
Who May Find This Useful
This discussion may be of interest to those studying nuclear physics, particularly in understanding nucleon behavior within atomic nuclei and the implications of nuclear forces on distribution patterns.