fyyfifowffums
- 2
- 0
Hi, I'm learning the basics of plasma assisted combustion so I've been reading up on a few articles related to it.
My question is regarding this article http://enu.kz/repository/2011/AIAA-2011-971.pdf
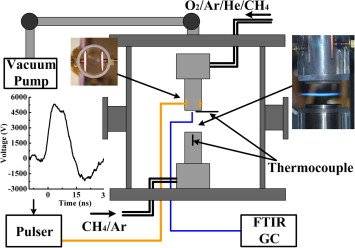
Using a setup consisting of a pair of counterflow burners, the composition of the oxidizer stream was fixed at O2/Ar/He/CH4(0.26:0.32:0.4:0.02) while the fuel stream was CH4 diluted by Ar (fuel mole fraction varied from 0.2 to 0.4). In this experiment, both oxidation and fuel reforming occurs. How do I find the equivalence ratio for both cases?
Oxidation: CH4 + 2O2→ CO2 + 2H2O
Fuel reforming: 2CH4 + O2→ 4H2 + 2CO
The equivalence ratio is defined as (fuel-to-oxidizer ratio)/ (fuel-to-oxidizer ratio)st and usually methane will be the fuel and oxygen as oxidizer, but in this case the oxidizer itself is premixed with methane so how do I calculate the equivalence ratio? Please help. I get so confused.
My question is regarding this article http://enu.kz/repository/2011/AIAA-2011-971.pdf
Using a setup consisting of a pair of counterflow burners, the composition of the oxidizer stream was fixed at O2/Ar/He/CH4(0.26:0.32:0.4:0.02) while the fuel stream was CH4 diluted by Ar (fuel mole fraction varied from 0.2 to 0.4). In this experiment, both oxidation and fuel reforming occurs. How do I find the equivalence ratio for both cases?
Oxidation: CH4 + 2O2→ CO2 + 2H2O
Fuel reforming: 2CH4 + O2→ 4H2 + 2CO
The equivalence ratio is defined as (fuel-to-oxidizer ratio)/ (fuel-to-oxidizer ratio)st and usually methane will be the fuel and oxygen as oxidizer, but in this case the oxidizer itself is premixed with methane so how do I calculate the equivalence ratio? Please help. I get so confused.