Homework Help Overview
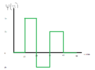
The discussion revolves around understanding the y-axis of a probability density graph, particularly in the context of quantum mechanics and wavefunctions. Participants are exploring the relationship between probability and probability density, and how to correctly interpret and manipulate these concepts mathematically.
Discussion Character
- Conceptual clarification, Assumption checking, Mathematical reasoning
Approaches and Questions Raised
- Participants are attempting to derive the correct probability density from a given wavefunction, questioning the validity of their initial assumptions about the graph's nature. There is discussion about the necessity of integrating the correct function and the implications of negative values in probability density.
Discussion Status
Some participants have identified misunderstandings regarding the nature of the graph and the definitions of probability versus probability density. There is ongoing exploration of how to properly convert probability into probability density, with some guidance offered on the need to square the wavefunction.
Contextual Notes
Participants are navigating constraints related to the definitions of probability density and the requirements for total probability, as well as the implications of negative values in their calculations.