Discussion Overview
The discussion revolves around naming hydrocarbons, specifically focusing on the correct nomenclature for various compounds as presented in an attached file. Participants explore concepts related to hydrocarbon chains, bond multiplicity, and the implications of structural diagrams on naming conventions.
Discussion Character
- Homework-related
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
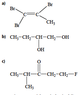
- One participant proposes a name for a compound as 1,1,2-tribromo-1-propene but expresses uncertainty about whether a CH3 group is a methyl group or part of the main chain.
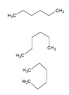
- Another participant questions how to consistently identify the main hydrocarbon chain in diagrams and asks which is longer between propane and ethane, noting bond multiplicity does not matter.
- Participants discuss bond multiplicity, with one noting that ethane is longer than propane but struggles to determine the placement of the CH3 group in the diagram.
- A participant explains that carbon chains are rarely straight and that single bonds allow for rotation, which may affect how groups are perceived in diagrams.
- One participant suggests that technically correct names for the compounds should differ from those initially proposed, citing historical naming conventions.
- There is confusion regarding the term "bond multiplicity," with participants seeking clarification on its meaning and relevance to naming hydrocarbons.
- Discussion includes the idea that adding a methyl group to a carbon chain results in a different compound, raising questions about naming conventions when side groups are involved.
- One participant asserts that if a methyl group is attached at the end of a chain, it is not considered a side group.
Areas of Agreement / Disagreement
Participants express varying levels of certainty regarding the correct naming of hydrocarbons, with some proposing alternative names while others defend their original choices. There is no consensus on the definitions and implications of bond multiplicity, nor on the naming conventions for compounds with methyl groups.
Contextual Notes
Some participants mention that the naming conventions discussed may be outdated or not aligned with current educational expectations, indicating potential limitations in the understanding of nomenclature among participants.
Who May Find This Useful
Students studying organic chemistry, particularly those focusing on hydrocarbon nomenclature and structural interpretation, may find this discussion relevant.