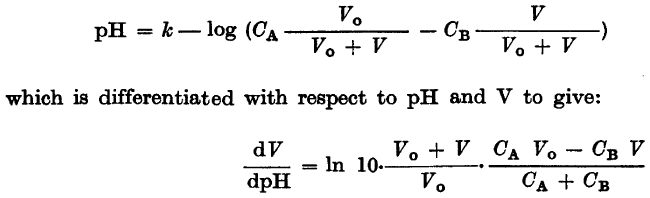Discussion Overview
The discussion revolves around the validity of taking the reciprocal in the differentiation of the relationship between pH and volume (V), specifically examining the expression dpH/dV. Participants explore different methods of differentiation and the implications of their approaches, including algebraic manipulation and the handling of logarithmic terms.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
- One participant questions the validity of taking the reciprocal after differentiating the equation for dpH/dV, expressing difficulty in achieving the same result through an alternative method of solving for V first.
- Another participant asserts that taking the reciprocal is valid, referencing the relationship between dy/dx and dx/dy.
- Participants discuss the algebraic manipulation of the original equation, suggesting that combining logarithmic terms may simplify the differentiation process.
- One participant shares their detailed differentiation steps, raising a question about a potential sign error in the expression for dpH/dV.
- Another participant reflects on their understanding of the equivalence point in the context of the pH versus V graph, indicating a realization about the relationship between the variables.
- There is mention of the Gran method and its relevance to the discussion, with participants expressing uncertainty about the algebraic inversion of the equation.
- Some participants express a desire to clarify their understanding and confirm their calculations, indicating ongoing uncertainty in the discussion.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the validity of taking the reciprocal in differentiation, as some support the approach while others express confusion and seek clarification. The discussion remains unresolved regarding the best method to differentiate and the implications of each approach.
Contextual Notes
Participants note potential complexities in the algebraic manipulation of logarithmic terms and the differentiation process, highlighting the challenges in achieving consistent results. There are also references to specific constants and terms that may require further clarification.
Who May Find This Useful
This discussion may be useful for students and professionals interested in the mathematical treatment of chemical equilibria, particularly in the context of pH and volume relationships, as well as those exploring differentiation techniques in applied mathematics.