tomdodd4598
- 137
- 13
Hey there,
I used Mathematica to find the (non-normalised) wave function of an electron in the vicinity of a Hydrogen nucleus, and converted the answer from one involving Whittaker functions to one involving generalised Laguerre polynomials. My result is shown below:
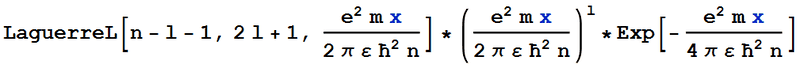
This agrees with the documented non-normalised wave function. However, when I multiply by the documented normalisation constant, to get the following wave function:
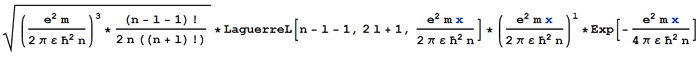
It is still not normalised - the integral from 0 to infinity of the absolute square is still not equal to 1.
Does anyone know what I'm doing wrong, and/or how to fix the wave function? Thanks in advance.
I used Mathematica to find the (non-normalised) wave function of an electron in the vicinity of a Hydrogen nucleus, and converted the answer from one involving Whittaker functions to one involving generalised Laguerre polynomials. My result is shown below:
This agrees with the documented non-normalised wave function. However, when I multiply by the documented normalisation constant, to get the following wave function:
It is still not normalised - the integral from 0 to infinity of the absolute square is still not equal to 1.
Does anyone know what I'm doing wrong, and/or how to fix the wave function? Thanks in advance.