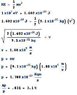SUMMARY
The discussion focuses on calculating the percentage uncertainty of momentum for a 1 keV electron when its position is known to within 1 Angstrom (Δx). Using the Heisenberg uncertainty principle, the uncertainty in momentum (Δp) is calculated as 5.275 x 10^-25 kg m/s. The kinetic energy equation is applied to determine the momentum (p) of the electron, resulting in a value of 1.708 x 10^-23 kg m/s. The final percentage uncertainty in momentum is determined to be 3.1%.
PREREQUISITES
- Understanding of the Heisenberg uncertainty principle
- Familiarity with kinetic energy equations
- Basic knowledge of electron properties (mass, charge)
- Ability to perform calculations involving scientific notation
NEXT STEPS
- Study the implications of the Heisenberg uncertainty principle in quantum mechanics
- Learn about the relationship between kinetic energy and momentum
- Explore advanced topics in quantum mechanics, such as wave-particle duality
- Investigate the behavior of electrons in various potential wells
USEFUL FOR
Students in physics, particularly those studying quantum mechanics, as well as educators and anyone interested in the principles of uncertainty in particle physics.