Discussion Overview
The discussion revolves around the potential to industrialize the photosynthesis process as a means to reduce atmospheric CO2 levels. Participants explore various aspects of this concept, including the feasibility, implications, and challenges associated with such an industrialization, as well as the broader environmental impacts.
Discussion Character
- Exploratory
- Debate/contested
- Technical explanation
- Mathematical reasoning
Main Points Raised
- Some participants question the completeness of our understanding of photosynthesis and its steps.
- There is a suggestion that while industrializing photosynthesis could produce O2, the management of the resulting carbon waste poses significant challenges.
- One participant argues that all agriculture can be seen as a form of industrialized photosynthesis and proposes using hardwood products to sequester carbon effectively.
- Concerns are raised about the sustainability of cutting down forests for hardwood and the implications of not allowing them to regrow.
- A participant presents calculations regarding CO2 emissions from gasoline and wood, questioning how much forest area would be needed to offset CO2 production and the practicalities of using the resulting wood.
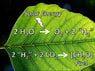
- There is mention of an artificial leaf technology that could potentially convert CO2 into usable fuel, with references to different versions of this technology.
Areas of Agreement / Disagreement
Participants express a range of views on the feasibility and implications of industrializing photosynthesis, with no clear consensus reached on the best approach or the practicality of the proposed solutions.
Contextual Notes
Participants highlight uncertainties regarding the efficiency of carbon absorption by trees and the complexities involved in managing biomass. There are also unresolved questions about the conversion of CO2 into usable products and the long-term sustainability of proposed methods.
Who May Find This Useful
This discussion may be of interest to those exploring environmental science, carbon capture technologies, sustainable forestry practices, and the industrial applications of photosynthesis.