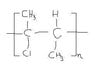
Polymerization of 2-Chloro-2-Butene: Solution
- Thread starter Macroer
- Start date
-
- Tags
- Polymer
Click For Summary
Discussion Overview
The discussion revolves around the polymerization of 2-chloro-2-butene through addition polymerization, focusing on the structure of the resulting polymer, the initiation and termination processes, and the representation of end groups in the polymer chain.
Discussion Character
- Homework-related
- Technical explanation
- Debate/contested
Main Points Raised
- One participant inquires about the appearance of the polymer formed from 2-chloro-2-butene and requests feedback on their proposed solution.
- Another participant asks for clarification on the structure of the initiator end of the molecule and how the polymerization process is terminated.
- A participant presents a proposed initiation step involving an initiator and the 2-chloro-2-butene molecule, indicating the formation of a radical.
- Concerns are raised about the accuracy of a drawing that represents 2-chloro-1-butene instead of the correct 2-butene version, with questions about the necessity of showing end groups.
- Further clarification is provided regarding the initiation step and the representation of end groups in the polymer structure.
- One participant expresses confusion about the presence of a second chlorine atom in the proposed structure.
- Another participant acknowledges the error regarding the chlorine atom and corrects the representation of the end groups in the polymer chain.
Areas of Agreement / Disagreement
Participants express differing views on the correct representation of the polymer structure, particularly regarding the end groups and the presence of chlorine atoms. The discussion remains unresolved as participants continue to clarify and challenge each other's claims.
Contextual Notes
There are uncertainties regarding the correct depiction of the polymer structure, the role of end groups, and the initiation process. Some assumptions about the chemical structures and reactions are not fully detailed, leading to potential misunderstandings.
Similar threads
- · Replies 0 ·
- · Replies 2 ·
- · Replies 1 ·
- · Replies 3 ·
- · Replies 2 ·
- · Replies 3 ·
- · Replies 11 ·