SUMMARY
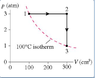
The discussion focuses on solving an isothermal process involving a monoatomic ideal gas, where heat is supplied at constant pressure and volume. The participant correctly identifies that the total work equals the total heat supplied, resulting in no change in internal energy due to the initial and final temperatures being the same. The relevant equation for work done during the process is W = nRT ln(V2/V1), which is crucial for calculating the heat supplied during the transitions from state 1 to state 2 (constant pressure) and from state 2 to state 4 (constant volume).
PREREQUISITES
- Understanding of ideal gas laws and properties
- Familiarity with the first law of thermodynamics
- Knowledge of isothermal processes and state variables
- Proficiency in using the equation W = nRT ln(V2/V1)
NEXT STEPS
- Study the concept of isothermal processes in thermodynamics
- Learn about the first law of thermodynamics and its applications
- Explore the relationship between pressure, volume, and temperature in ideal gases
- Investigate the implications of constant pressure and constant volume on heat transfer
USEFUL FOR
Students studying thermodynamics, particularly those focusing on gas laws and isothermal processes, as well as educators seeking to clarify these concepts for their students.