nhrock3
- 403
- 0
the standard potential for
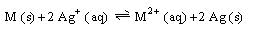
is 1.081
what is the standard potential for M
?
is 1.081
what is the standard potential for M
?
The standard potential for M is established at 1.081 volts. This value is crucial for understanding the electrochemical series and its applications in various chemical reactions. The total potential is directly related to the standard potentials of half-cells, which are essential for calculating cell voltages in electrochemical cells. Additionally, the standard potential of the silver half-cell is a key reference point in electrochemistry.
PREREQUISITESChemistry students, electrochemists, and professionals involved in battery technology or corrosion science will benefit from this discussion.