gangsterlover
- 31
- 0
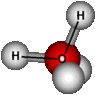
What role do the 2 lone electron pairs have in a h2o molecule when hydrogen and oxygen bond covalently. I`ve understood that the oxygen side of the h2o molecule is negative because it is more electronegative and the elctrons spend more time on that "side". While the hydrogen "side" is more positive beaause the electrons spend less time there. However, everytime I look at an h2o graphical representation of an h2o molecule I never see the hybridized sp3 orbitals that have been formed for the 2 lone electron pairs on oxygen. Where are they. I`ve read that h2o is tetrahedral. Is that wrong? Also, if the 2 lone electron pairs are on the oxygen side then they would just increase the negative polarity of the oxygen "side".
Right, wrong?
Thanks,
Right, wrong?
Thanks,