- #1
jadelamlam
- 51
- 0
Homework Statement
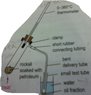
My textbook states that one of the safety precautions to be taken in the fractional distillation of petroleum(in school laboratory) is using a small flame to heat the rocksil(rocksil is soaked with petroleum),and avoil using stationary flame for heating.
Can anyone explain?
Homework Equations
The Attempt at a Solution
I guess the reason of using a small flame is to avoid the breaking of test tube(test tube is used to hold the rocksil)?But I have no idea on why shouldn't we use a stationary flame for heating.