- #1
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
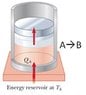
Why is this ISOTHERMAL and not ISOBARIC?
- Thread starter Farina
- Start date
-
- Tags
- Isobaric Isothermal
In summary, this is an Ideal Gas process. The pressure is such that the weight of the piston balances it, so the piston will not begin to move and the gas will not expand.
Science news on Phys.org
- #2
Haborix
- 339
- 374
Hello,
Quick question. Does the piston contain an ideal gas? If so, remember what an isotherm looks like on P vs. V diagram for an ideal gas.
I hope this helps. If not, then I'm sure someone else will come along and help you sort it out.
Quick question. Does the piston contain an ideal gas? If so, remember what an isotherm looks like on P vs. V diagram for an ideal gas.
I hope this helps. If not, then I'm sure someone else will come along and help you sort it out.
- #3
Farina
- 39
- 0
It is an Ideal Gas - thank you. I definitely see how it's an isothermal process (due to the heat reservoir). I'm still not, however, seeing how it is also not an isobaric process.
- #4
nasu
Homework Helper
- 4,416
- 884
What makes the piston to move up?
You did not describe the actual mechanism illustrated here.
You did not describe the actual mechanism illustrated here.
- #5
Farina
- 39
- 0
According to the textbook, this is simply a Carnot process (see attached image). My point is this: if the piston is free to move, then it will move however it needs to move to maintain a constant pressure - at least this is how I'm used to hearing the description of an isobaric process.
So, again, isn't this BOTH an isobaric process and isothermal process?
This seems to be quite a brain teaser.
So, again, isn't this BOTH an isobaric process and isothermal process?
This seems to be quite a brain teaser.
Attachments
- #6
nasu
Homework Helper
- 4,416
- 884
I think I understand your confusion.
You assume that the pressure of the gas is such that balances the weight of the piston.
If this were the case, the piston will not begin to move and the gas will not expand. And no heat will be absorbed, as the gas has the same temperature as the reservoir, in state A.
Imagine that the pressure in state A is larger than the weight of the piston divided by its area.
The gas pushes the piston and expands, lowering its pressure in the process. To keep its temperature constant during expansion it absorbs heat from the reservoir.
You assume that the pressure of the gas is such that balances the weight of the piston.
If this were the case, the piston will not begin to move and the gas will not expand. And no heat will be absorbed, as the gas has the same temperature as the reservoir, in state A.
Imagine that the pressure in state A is larger than the weight of the piston divided by its area.
The gas pushes the piston and expands, lowering its pressure in the process. To keep its temperature constant during expansion it absorbs heat from the reservoir.
- #7
Farina
- 39
- 0
Ah, yes - you're right, that's what I was missing. Thank you very much!
- #8
nasu
Homework Helper
- 4,416
- 884
You are very welcome.
1. Why is this process considered isothermal and not isobaric?
This process is considered isothermal because the temperature of the system remains constant throughout the process. Isobaric processes, on the other hand, have a constant pressure throughout the process.
2. How can you determine if a process is isothermal or isobaric?
A process is considered isothermal if the temperature remains constant, while it is considered isobaric if the pressure remains constant.
3. Why is it important to differentiate between isothermal and isobaric processes?
It is important to differentiate between these two processes because they have different effects on the system. An isothermal process does not change the internal energy of the system, while an isobaric process can change the internal energy by doing work on or by adding heat to the system.
4. What type of systems are most likely to undergo an isothermal process?
Ideal gases are most likely to undergo an isothermal process because they have no intermolecular forces and thus no change in temperature when the pressure changes. Real gases may also undergo an isothermal process under certain conditions.
5. Can a process be both isothermal and isobaric?
Yes, a process can be both isothermal and isobaric. This would occur when the temperature and pressure both remain constant throughout the process.
Similar threads
-
Thermodynamics
- Replies
- 22
- Views
- 2K
-
Thermodynamics
- Replies
- 11
- Views
- 326
-
Thermodynamics
- Replies
- 11
- Views
- 5K
-
Thermodynamics
- Replies
- 12
- Views
- 1K
- Replies
- 12
- Views
- 2K
- Replies
- 3
- Views
- 1K
-
Thermodynamics
- Replies
- 1
- Views
- 653
- Replies
- 81
- Views
- 3K
-
Introductory Physics Homework Help
- Replies
- 3
- Views
- 128
-
Thermodynamics
- Replies
- 2
- Views
- 1K
Share: