Discussion Overview
The discussion revolves around the reaction of 2-butene with hydrogen chloride gas, contrasting it with the reaction of 1-butene. Participants explore the reasons why only one product is formed from 2-butene while two products are typically produced from 1-butene, delving into concepts of organic chemistry and reaction mechanisms.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
- Mathematical reasoning
Main Points Raised
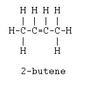
- One participant notes that 2-butene reacts with HCl to form 2-chlorobutane, questioning the formation of two products from 1-butene.
- Another participant suggests drawing the products of both reactions to clarify the distinct products formed.
- A participant corrects a previous typo regarding the reactant, confirming that HCl is the correct reagent.
- A further explanation is provided regarding the attachment of H and Cl to the carbons in 2-butene, leading to the possibility of two distinct products based on the attachment positions.
- Markovnikov's rule is mentioned as a relevant concept for understanding product formation, although its applicability to the specific problem is debated.
- One participant expresses uncertainty about the actual mechanism of the reaction and the distribution of products in the case of 1-butene.
Areas of Agreement / Disagreement
Participants express differing views on the nature of the products formed from 1-butene, with some asserting that two distinct products are produced while others question the implications of this. The discussion remains unresolved regarding the specifics of the reaction mechanisms and product distributions.
Contextual Notes
Participants acknowledge the complexity of the reaction mechanisms and the potential for different interpretations based on the course scope and instructor expectations. There are also mentions of pending visual aids that may clarify the discussion.