Discussion Overview
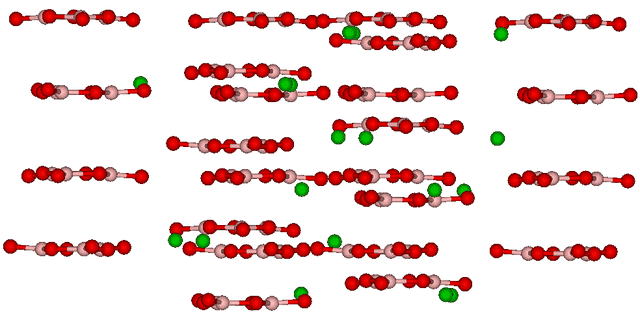
The discussion revolves around the structure and properties of beta barium borate (BBO) crystals, specifically focusing on the chemical composition and the arrangement of elements within the crystal structure. Participants express curiosity about the nature of these crystals and their formation, drawing from their backgrounds in chemistry.
Discussion Character
- Exploratory
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- One participant notes a discrepancy between the chemical formula BaB2O4 and the visual representation of the crystal, highlighting the colors associated with each element.
- Another participant emphasizes that the stoichiometric formula indicates relative amounts of elements but does not provide information about the crystal structure, questioning if there is doubt about the proportions of elements.
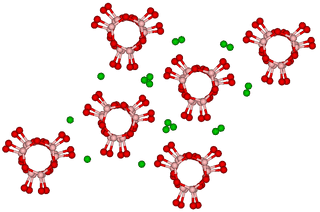
- A participant shares their background in organic chemistry and expresses curiosity about how barium interacts with boron and oxygen to form a ring structure, questioning the role of barium as a spectator cation in solid-state formation and the arrangement of these units in triplets.
- Another participant challenges the assumption that rings of BO3 units do not occur in solution, suggesting a potential overlap in behavior between solid and aqueous states.
Areas of Agreement / Disagreement
Participants do not reach a consensus, as there are competing views regarding the structural characteristics of BBO crystals and the behavior of BO3 units in different environments.
Contextual Notes
The discussion includes assumptions about the chemical behavior of barium, boron, and oxygen, as well as the structural implications of their interactions, which remain unresolved.
Who May Find This Useful
Individuals interested in crystal chemistry, materials science, or those studying the properties of barium borate and its applications in various fields may find this discussion relevant.