hsbc2003
- 5
- 0
Please add this title: Calculating the mass and number of atoms of a structural I beam.
A structural I beam is made of iron> A view of its cross-section and its dimensions is shown in the figure, where d = 2 cm, w= 31 cm, h= 28 cm and the length (not shown) of the beam is L= 1.53 m.
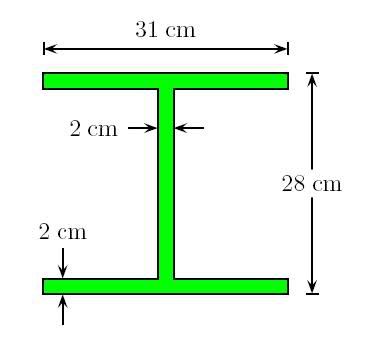
The density of iron is 7560 kg/m^3. The atomic weight of iron is 55.85 g/mol and Avagadro number is Na= 6.02214e23/mol.
What is the mass of a section 1.53 m long? (in kg)
How many atoms are there in this section? (in units of atoms)
I calculated the volume of the I beam ( (h*w*l)-(the volume of the empty space))
and then I multiply it by the density of iron to get the mass.
but the answer was wrong, thus i I tried to calculate the number of atoms using Avagadro's number the answer will be wrong as well...
I hope you help me calculating the mass...
Thank you
Homework Statement
A structural I beam is made of iron> A view of its cross-section and its dimensions is shown in the figure, where d = 2 cm, w= 31 cm, h= 28 cm and the length (not shown) of the beam is L= 1.53 m.
The density of iron is 7560 kg/m^3. The atomic weight of iron is 55.85 g/mol and Avagadro number is Na= 6.02214e23/mol.
What is the mass of a section 1.53 m long? (in kg)
How many atoms are there in this section? (in units of atoms)
The Attempt at a Solution
I calculated the volume of the I beam ( (h*w*l)-(the volume of the empty space))
and then I multiply it by the density of iron to get the mass.
but the answer was wrong, thus i I tried to calculate the number of atoms using Avagadro's number the answer will be wrong as well...
I hope you help me calculating the mass...
Thank you
Last edited by a moderator: