lumealume
- 1
- 0
Hi! I had this problem for homework (Mastering Physics), and I'm not sure what I'm doing incorrectly. Is there something fundamental that I'm misunderstanding? Each time I do this, I get 0.717136.. T as my solution
1. Homework Statement
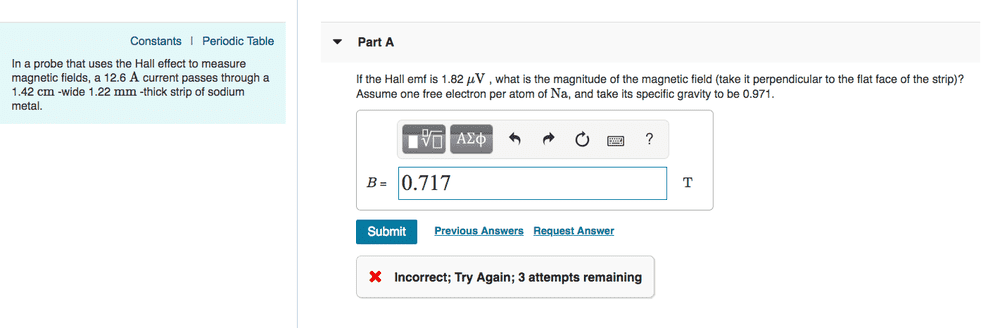
I = 12.6 A
w = 0.0142 m
t = 0.00122 m
E_H = 1.82 * 10^(-6) V
G_s = 0.971
B = ?
1/(qn) = Vt/(IB)


EDIT: (editing the post currently because I just realized that I shouldn't just post images. I'm new here!)
EDIT2: Finished editing post with thought process
1. Homework Statement
I = 12.6 A
w = 0.0142 m
t = 0.00122 m
E_H = 1.82 * 10^(-6) V
G_s = 0.971
B = ?
Homework Equations
1/(qn) = Vt/(IB)
The Attempt at a Solution
- I have the formula for B from 1/(qn) = Vt/(IB) => B = Vtqn/I
- Plug V, t, q (electron charge), and I into get B = (1.82 * 10^(-6) V)(0.00122 m)(1.6 * 10^(-19)) * n / 12.6 A
- Now it's time to solve for n: G_s = 0.971 = (Density of Na)/(Density of water) = (Density of Na)/(1000 kg/m^3)
- Density of Na = 971 kg/m^3
- I want n = electron density in units of electrons / m^3.
- There is 1 electron per 1 atom of Na so...
- n = Density of Na * mols of Na per mass in kg * Na atoms per mol * electron per Na atom will cancel everything out nicely to get me electrons / m^3
- n = (971 kg/m^3) * (mol/(0.02299kg)) * (6.022*10^23 atoms of Na / mol) * (1 electron / 1 atom Na)
- Putting it all together: B = (1.82 * 10^(-6) V)(0.00122 m)(1.6 * 10^(-19)) * (971 kg/m^3) * (mol/(0.02299kg)) * (6.022*10^23 atoms of Na / mol) * (1 electron / 1 atom Na) / 12.6 A
- B = 0.717 T
EDIT: (editing the post currently because I just realized that I shouldn't just post images. I'm new here!)
EDIT2: Finished editing post with thought process
Attachments
Last edited:

