Discussion Overview
The discussion revolves around calculating the specific heat and latent heats of fusion for a solution of NaCl in water (H2O), particularly focusing on the effects of concentration on these properties. Participants explore theoretical aspects, mathematical formulations, and practical implications of these calculations.
Discussion Character
- Exploratory
- Technical explanation
- Mathematical reasoning
- Homework-related
Main Points Raised
- Some participants inquire whether it is possible to calculate the latent heats of fusion and specific heat for a NaCl and H2O solution, particularly at different concentrations.
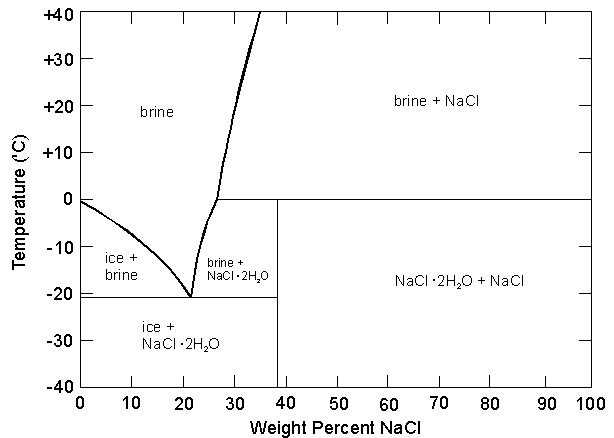
- One participant suggests that the heat of fusion will vary with the concentration of the brine.
- Another participant questions if there would be different heats of fusion at specific temperatures for a 10% NaCl solution.
- It is noted that at a 10% solution, no NaCl formation occurs at certain temperatures, and the heat of fusion is relevant only along the line between brine and brine + NaCl.
- Participants discuss the behavior of the solution as it cools towards the eutectic point and the implications for calculating latent heat of fusion.
- One participant provides a detailed mathematical approach using Raoult's law and chemical potential to derive a formula related to freezing point depression.
- Another participant expresses difficulty in resolving a specific problem and requests an example to clarify the calculations.
- There is mention of converting weight percent to molar fraction as a necessary step in calculations.
- A participant shares a derived formula for freezing point depression and expresses intent to conduct a practical experiment.
- One participant presents a calculation for specific heat based on their derived values but questions the validity of the resulting large number.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the calculations or the implications of their findings. Multiple competing views and approaches remain, particularly regarding the effects of concentration on latent heat and specific heat.
Contextual Notes
Some calculations depend on specific assumptions about concentration and temperature, and participants highlight the need for careful consideration of these factors in their discussions.