SUMMARY
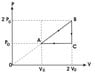
The discussion focuses on the calculation of the income from an ideal gas adiabatic coefficient γ during the ABCA cycle, as depicted in the referenced figure. Participants are tasked with demonstrating that the income is less than the yield of a Carnot cycle operating between the same temperature extremes. The equation P=nRT/V is utilized to derive the necessary calculations, and the accuracy of the work done in the cycle is confirmed. Clarification is sought regarding the specific heat Q calculated in the solution attempt.
PREREQUISITES
- Understanding of thermodynamic cycles, specifically the ABCA cycle.
- Familiarity with the Carnot cycle and its efficiency principles.
- Knowledge of the ideal gas law, particularly P=nRT/V.
- Basic concepts of adiabatic processes in thermodynamics.
NEXT STEPS
- Study the principles of the Carnot cycle and its efficiency calculations.
- Explore the derivation and implications of the ideal gas law in thermodynamic processes.
- Investigate adiabatic processes and their characteristics in thermodynamics.
- Learn about the significance of heat transfer in thermodynamic cycles.
USEFUL FOR
Students and professionals in thermodynamics, mechanical engineers, and anyone involved in the study of heat engines and energy efficiency.