gfd43tg
Gold Member
- 949
- 48
Hello,
I have the problem statement attached in the thumbnail. I am confused about this question. I looked at the solution manual, and they are saying that the pressure at states 3 and 4 are the same, as well as at states 1 and 2. I don't see how this is. Looking at the carnot cycle PV diagram, clearly all 4 states are at different pressures.
Anyways, I did the calculation with the same pressures. Now I am working on part C, and I thought since step 1-->2 is an isotherm, I could use the equation Q=RT ln(P2/P1). However, if P2 and P1 are the same, then Q is zero. What is going on here?

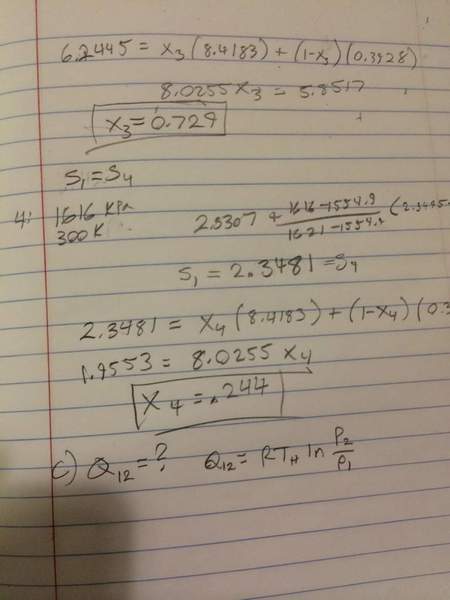
I have the problem statement attached in the thumbnail. I am confused about this question. I looked at the solution manual, and they are saying that the pressure at states 3 and 4 are the same, as well as at states 1 and 2. I don't see how this is. Looking at the carnot cycle PV diagram, clearly all 4 states are at different pressures.
Anyways, I did the calculation with the same pressures. Now I am working on part C, and I thought since step 1-->2 is an isotherm, I could use the equation Q=RT ln(P2/P1). However, if P2 and P1 are the same, then Q is zero. What is going on here?
Attachments
Last edited:
