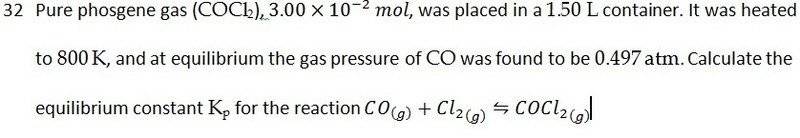Discussion Overview
The discussion revolves around determining the partial pressure of gases at equilibrium, specifically focusing on the stoichiometric relationships in chemical reactions involving phosgene, carbon monoxide, and chlorine. Participants explore the reasoning behind calculating partial pressures and the implications of stoichiometric ratios in equilibrium conditions.
Discussion Character
- Technical explanation
- Debate/contested
Main Points Raised
- One participant suggests that to find the partial pressure of phosgene, one must subtract the pressure of CO from the initial pressure, questioning the reasoning behind this approach.
- Another participant argues that the stoichiometric ratios are not 1:1:1, implying that this affects the equilibrium constant and the interpretation of the stoichiometric equation.
- A participant expresses confusion about the stoichiometric ratios, asserting that the balanced equation indicates a 1:1:1 relationship among the gases involved.
- In a related question, a participant notes that the equilibrium pressures add up to more than 100%, questioning how this is possible given the dissociation percentages of the gases.
- Another participant reiterates the confusion regarding the stoichiometric ratios, emphasizing that the reaction produces different amounts of each gas, which contradicts the 1:1:1 assumption.
Areas of Agreement / Disagreement
Participants express disagreement regarding the interpretation of stoichiometric ratios and their implications for calculating partial pressures. There is no consensus on the correct reasoning behind the calculations or the relationships between the gases at equilibrium.
Contextual Notes
Participants highlight the complexity of interpreting stoichiometric relationships in equilibrium situations, indicating that assumptions about gas ratios may lead to confusion in calculations. The discussion remains open-ended with unresolved questions about the mathematical relationships involved.