Discussion Overview
The discussion revolves around the interpretation of photospectrometry results for a dye-doped thin film. Participants explore the implications of absorbance measurements, sample preparation, and the materials used in the experiment, focusing on both theoretical and practical aspects of spectroscopy.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Experimental/applied
Main Points Raised
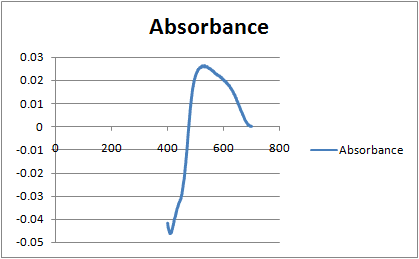
- One participant notes the presence of negative absorbance, suggesting potential issues with calibration or external light interference.
- Another participant points out the low absorbance value of 0.03 AUFS, questioning whether the spectrophotometer was properly zeroed and inquiring about the solvent used.
- There is a repeated emphasis on the need for proper sample handling, including whether the sample is solid or liquid and the type of cuvette used.
- Concerns are raised about potential fluorescence from the glass slide or the sample itself, which could affect the measurements.
- Recommendations are made to use quartz cuvettes for better results, with suggestions to increase dye concentration for more accurate quantification.
- Questions are posed regarding the effects of orthosilicates and phosphates on the UV-Vis spectrum, particularly in relation to the dye's behavior in different solvent mixtures.
Areas of Agreement / Disagreement
Participants express various viewpoints on the interpretation of the spectrometry results, with no clear consensus on the best practices for sample preparation or the implications of the observed data. Multiple competing views remain regarding the effects of different materials and methods on the measurements.
Contextual Notes
Participants mention potential limitations related to sample preparation, calibration procedures, and the choice of solvents, but these aspects remain unresolved and are subject to further exploration.
Who May Find This Useful
This discussion may be useful for researchers and practitioners involved in spectroscopy, particularly those working with dye-doped materials or thin films in experimental settings.