Discussion Overview
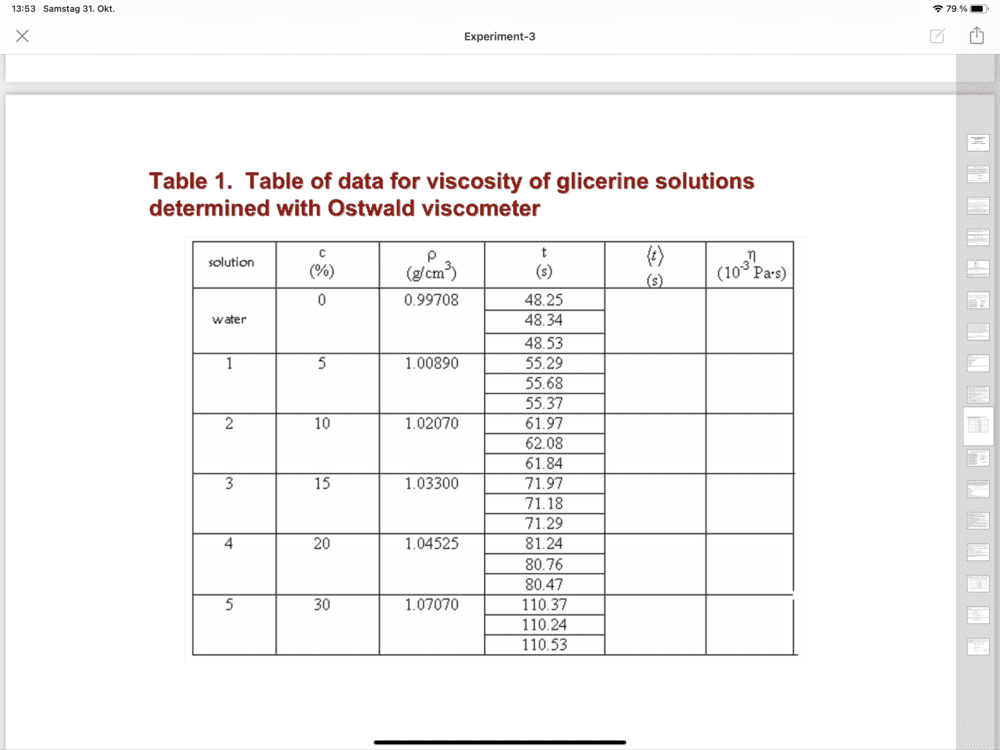
The discussion revolves around the use of an Ostwald viscometer for measuring the viscosity of water at different temperatures. Participants are seeking clarification on the relevant formulas, units, and how to interpret the data collected during the experiment.
Discussion Character
- Homework-related
- Technical explanation
- Conceptual clarification
Main Points Raised
- One participant questions the meaning of "h2o" and its relation to viscosity measurements, specifically asking if 0.99708 refers to water's density.
- Another participant expresses confusion about the original question, suggesting a misunderstanding of the relationship between the formula, the substance, and its properties.
- A participant mentions the need for the viscosity of water at 0°C and suggests using a specific reference for this value from the CRC handbook.
- There is a discussion about converting viscosity values from centipoise to Pascal seconds, with one participant asking for clarification on the units involved.
- Participants discuss the average time taken for water to pass through the viscometer and its relevance to viscosity calculations, with one participant noting the importance of averaging multiple trials.
Areas of Agreement / Disagreement
Participants express varying levels of understanding regarding the formulas and units used in the experiment. There is no consensus on the correct interpretation of certain values or the methodology for calculating viscosity.
Contextual Notes
Some participants mention specific temperature values and viscosity units, but there is uncertainty about the correct conversions and the sources of the data being used. The discussion reflects a lack of clarity on how to apply the Ostwald formula correctly.
 48,25
48,25



