ramzerimar
- 178
- 23
Someone showed this to me, and I'm struggling to explain why this perpetual motion scheme is impossible.
A picture:
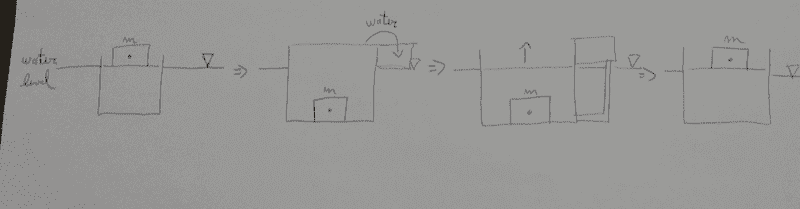
Basically, this is a cylinder within a large reservoir, with water at the level of the reservoir water level. The mass would be dropped and then reach the bottom of the cylinder, and the overflowing water could be collected at the top. This water is now at a higher level than the large reservoir water level (and the water inside the cylinder could be maintained at the reservoir water level by the opening of some kind of valve). The higher pressure at this column of collected water would be used to bring the mass back to its original position (by means of a hydraulic actuator at the bottom, or something like that).
I'm neglecting all dissipative effects here. In the ideal case, does this break the first law of thermodynamics? My analysis is that the energy is conserved, since the mass "m" has some initial potential energy at the start and is back to that same energy level at the end of the cycle. Or does it break the second law because entropy increases?
A picture:
Basically, this is a cylinder within a large reservoir, with water at the level of the reservoir water level. The mass would be dropped and then reach the bottom of the cylinder, and the overflowing water could be collected at the top. This water is now at a higher level than the large reservoir water level (and the water inside the cylinder could be maintained at the reservoir water level by the opening of some kind of valve). The higher pressure at this column of collected water would be used to bring the mass back to its original position (by means of a hydraulic actuator at the bottom, or something like that).
I'm neglecting all dissipative effects here. In the ideal case, does this break the first law of thermodynamics? My analysis is that the energy is conserved, since the mass "m" has some initial potential energy at the start and is back to that same energy level at the end of the cycle. Or does it break the second law because entropy increases?