Discussion Overview
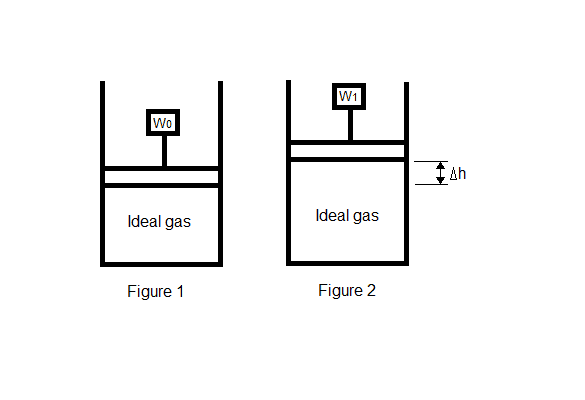
The discussion revolves around the calculation of work done in a system where mass and temperature change, particularly in the context of a piston mechanism. Participants explore the implications of different thermodynamic processes, such as isothermal and adiabatic conditions, and how these affect the equations used to calculate work.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- One participant presents an equation for work that includes terms for both heat capacity and gravitational potential energy, suggesting a combined approach to calculating work when mass and temperature change.
- Another participant requests clarification on the approach taken to solve the problem, indicating that understanding the reasoning behind the equations is essential for meaningful answers.
- A question is raised regarding whether the process is isothermal or adiabatic, highlighting the importance of the thermodynamic context in the calculations.
- It is noted that the use of the constant pressure heat capacity (C[P]) implies an assumption of constant pressure, which may influence the interpretation of the equations presented.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the nature of the thermodynamic process involved, with differing views on whether it is isothermal or adiabatic. The discussion remains unresolved regarding the implications of these conditions on the work calculations.
Contextual Notes
The discussion lacks detailed explanations of the assumptions behind the equations, such as the specific conditions under which they apply. There is also ambiguity regarding the definitions of the variables used in the equations, which may affect the clarity of the discussion.
Who May Find This Useful
This discussion may be useful for individuals interested in thermodynamics, particularly those exploring the relationship between work, mass, and temperature changes in mechanical systems.