Homework Help Overview
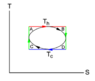
The discussion revolves around calculating the efficiency of a van der Waals gas cycle represented on a temperature-entropy (T-S) diagram. The original poster presents a problem involving n moles of gas following the van der Waals equation and seeks to determine the efficiency of a circular cycle parameterized by temperatures T_h and T_c.
Discussion Character
- Exploratory, Conceptual clarification, Problem interpretation
Approaches and Questions Raised
- The original poster attempts to calculate the efficiency by integrating over curves on the T-S diagram to find heat inputs and outputs. Some participants question the representation of the cycle as a circle due to differing units of temperature and entropy, while others seek clarification on deriving expressions for temperature as a function of entropy.
Discussion Status
Participants are actively exploring the implications of the problem setup, with some providing insights into the representation of the cycle. There is an acknowledgment of the challenges in calculating the necessary integrals and the need for a proper expression relating T and S. No consensus has been reached regarding the approach to the efficiency calculation.
Contextual Notes
Participants note that the task specifies a circular representation on the T-S diagram, which raises questions about the validity of this representation given the differing units of temperature and entropy. The original poster expresses uncertainty about how to proceed with the calculations based on the van der Waals equation.