SUMMARY
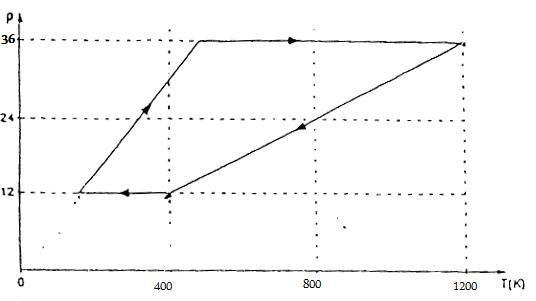
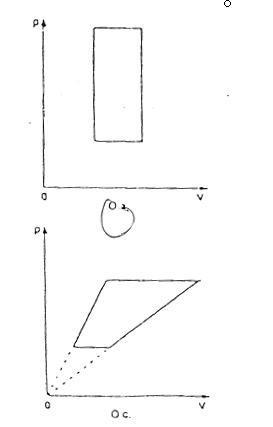
An ideal gas undergoes a cycle process represented in a PV graph, demonstrating the relationship between pressure (p), volume (V), and temperature (T) as defined by the equation pV = nRT. The upper graph illustrates linear relationships between temperature and pressure, indicating constant volume, which results in vertical lines in the p-V graph. In contrast, the lower graph fails to meet these criteria, as it does not maintain the necessary conditions for an ideal gas during the cycle process.
PREREQUISITES
- Understanding of the ideal gas law (pV = nRT)
- Familiarity with PV diagrams and their interpretation
- Knowledge of thermodynamic processes and cycles
- Basic principles of gas behavior under varying conditions
NEXT STEPS
- Study the implications of the ideal gas law in different thermodynamic processes
- Explore the characteristics of isothermal and adiabatic processes in PV diagrams
- Learn about the Carnot cycle and its significance in thermodynamics
- Investigate real gas behavior and deviations from ideal gas laws
USEFUL FOR
Students of thermodynamics, physics enthusiasts, and professionals in engineering fields who require a deeper understanding of gas behavior in cycle processes.