Flucky
- 93
- 1
Edit: well this is frustrating, not sure why the itex things aren't working..
Edit2: I've attached a picture of the question at the bottom as well.
The cohesive energy of a solid noble gas may be written as
(See picture below)
Derive an expression for the equilibrium separation r0 of the atoms.
At Ecohesive, r=r0 and dE/dr0 = 0
So first I rearranged the main equation into something nicer to differentiate:
E = 6.065Br[itex]^{-12}_{0}[/itex] - 7.225Ar[itex]^{-6}_{0}[/itex]
Then differentiated:
dE/dr0 = -72.78[itex]\frac{B}{r^{13}_{0}}[/itex] + 43.35[itex]\frac{A}{r^{7}_{0}}[/itex] = 0
43.35[itex]\frac{A}{r^{7}_{0}}[/itex] = 72.78[itex]\frac{B}{r^{13}_{0}}[/itex]
r[itex]^{6}_{0}[/itex] = [itex]\frac{72.78}{43.35}[/itex][itex]\frac{B}{A}[/itex]
So r0 = ([itex]\frac{72.78}{43.35}[/itex][itex]\frac{B}{A}[/itex])[itex]\frac{1}{6}[/itex]
But this looks pretty horrible, is it any good?
Cheers.
-------------------------------------------------------------------------------------------
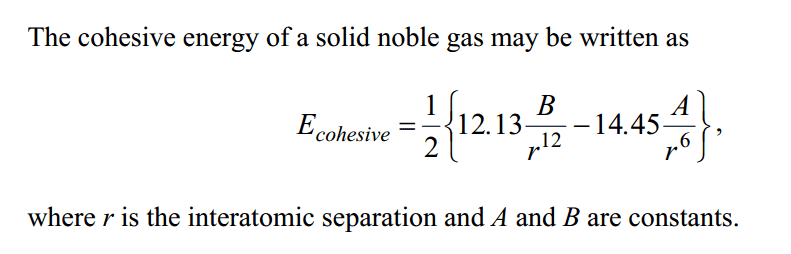
Edit2: I've attached a picture of the question at the bottom as well.
Homework Statement
The cohesive energy of a solid noble gas may be written as
(See picture below)
Derive an expression for the equilibrium separation r0 of the atoms.
The Attempt at a Solution
At Ecohesive, r=r0 and dE/dr0 = 0
So first I rearranged the main equation into something nicer to differentiate:
E = 6.065Br[itex]^{-12}_{0}[/itex] - 7.225Ar[itex]^{-6}_{0}[/itex]
Then differentiated:
dE/dr0 = -72.78[itex]\frac{B}{r^{13}_{0}}[/itex] + 43.35[itex]\frac{A}{r^{7}_{0}}[/itex] = 0
43.35[itex]\frac{A}{r^{7}_{0}}[/itex] = 72.78[itex]\frac{B}{r^{13}_{0}}[/itex]
r[itex]^{6}_{0}[/itex] = [itex]\frac{72.78}{43.35}[/itex][itex]\frac{B}{A}[/itex]
So r0 = ([itex]\frac{72.78}{43.35}[/itex][itex]\frac{B}{A}[/itex])[itex]\frac{1}{6}[/itex]
But this looks pretty horrible, is it any good?
Cheers.
-------------------------------------------------------------------------------------------
Last edited: