AN630078
- 242
- 25
- Homework Statement
- Hello, I have been reviewing the photoelectric effect when I came across the admittedly rather long problem below. I was keen to try to solve it since I had not used a graphical analysis for the photoelectric effect before, e.g. finding the work function in this way which has lead to some minor confusion. Could anyone offer any advice if I can improve upon my answers here, or generally dispense some light on the topic ?
Different stopping potentials were measured for different frequencies of electromagnetic radiation.
1. a. Plot the results from table 1 in a graph with maximum kinetic energy EK on the y-axis and frequency f on the x-axis.
b. Give the value of:
i. The threshold frequency
ii. The work function
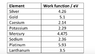
c. Using Table 2 and your answer to b ii, find the metal that is being tested.
d. Using your graph and Table 3, approximately how much kinetic energy would an electron
emitted from an incident blue photon have?
e. What would happen if a beam of yellow light was shone on the metal?
- Relevant Equations
- 1/2 m v^2 max = hf-Φ
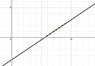
1. a)I have plotted the graph on desmos and attached an image here.
b i. The threshold frequency is equal to the x-intercept ~ 5.6*10^14 Hz
ii. The work function is equal to the y-intercept ~ -3.75*10^19 J (would it be correct to state that this value is negative?)
c. Convert to eV;
3.75*10^19 J=2.34375 eV
Evaluating table 2 I believe that this approximately corresponds to the work function of sodium, 2.36 eV. Such differences in values may have arisen from graphical uncertainties on my part.
d. The photoelectric effect equation states;
1/2 m v^2 max = hf-Φ
Evaluating table 3 a blue photon has a frequency in range 606-668 THz. Converting to Hz this 6.06*10^14 Hz-6.68*10^14 Hz
Would the work function here be that I found with my graph ~ 3.75*10^19 J ?
1/2 m v^2 max =6.63*10^-34* 6.06*10^14-3.75*10^19
1/2 m v^2 max = -3.75 *10^19 J
1/2 m v^2 max =6.63*10^-34* 6.68*10^14-3.75*10^19
1/2 m v^2 max = -3.75 *10^19J
Which is the same for both extremes of the range of the blue photon's frequency. Would the value of the work function here be negative so the actual kinetic energy emitted from an incident blue photon would be positive i.e. 3.75 *10^19J?
e. If a beam of yellow light was shone on the metal the energy of the photon can be found by (frequency range of a yellow photon is 5.08*10^14 Hz-5.26*10^14
E=hf=6.63*10^-34*5.08*10^14=3.368*10^19
E=6.63*10^-34*5.26*10^14=3.487*10^19
Convert to eV; Energy in range= 2.10-2.12 eV
The work function of the sodium has been shown to be 2.36 in table 2. Since this energy is less than the work function, meaning the yellow photon does not have sufficient energy to eject an electron from the sodium metal.
b i. The threshold frequency is equal to the x-intercept ~ 5.6*10^14 Hz
ii. The work function is equal to the y-intercept ~ -3.75*10^19 J (would it be correct to state that this value is negative?)
c. Convert to eV;
3.75*10^19 J=2.34375 eV
Evaluating table 2 I believe that this approximately corresponds to the work function of sodium, 2.36 eV. Such differences in values may have arisen from graphical uncertainties on my part.
d. The photoelectric effect equation states;
1/2 m v^2 max = hf-Φ
Evaluating table 3 a blue photon has a frequency in range 606-668 THz. Converting to Hz this 6.06*10^14 Hz-6.68*10^14 Hz
Would the work function here be that I found with my graph ~ 3.75*10^19 J ?
1/2 m v^2 max =6.63*10^-34* 6.06*10^14-3.75*10^19
1/2 m v^2 max = -3.75 *10^19 J
1/2 m v^2 max =6.63*10^-34* 6.68*10^14-3.75*10^19
1/2 m v^2 max = -3.75 *10^19J
Which is the same for both extremes of the range of the blue photon's frequency. Would the value of the work function here be negative so the actual kinetic energy emitted from an incident blue photon would be positive i.e. 3.75 *10^19J?
e. If a beam of yellow light was shone on the metal the energy of the photon can be found by (frequency range of a yellow photon is 5.08*10^14 Hz-5.26*10^14
E=hf=6.63*10^-34*5.08*10^14=3.368*10^19
E=6.63*10^-34*5.26*10^14=3.487*10^19
Convert to eV; Energy in range= 2.10-2.12 eV
The work function of the sodium has been shown to be 2.36 in table 2. Since this energy is less than the work function, meaning the yellow photon does not have sufficient energy to eject an electron from the sodium metal.