SUMMARY
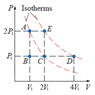
The discussion focuses on calculating the work done on a monatomic ideal gas transitioning from state A to state D via a PV diagram, specifically through a constant pressure path A-E and a constant temperature path E-D. The user initially calculated the work done using the formula W = P_i(V_f - V_i) but questioned the correctness of their approach, particularly regarding the pressure values used for each segment of the process. The correct interpretation of pressure during isothermal processes and the application of the first law of thermodynamics are critical to accurately determining work, internal energy change, and heat flow.
PREREQUISITES
- Understanding of the first law of thermodynamics
- Knowledge of PV diagrams and their interpretation
- Familiarity with monatomic ideal gas behavior
- Proficiency in calculating work done in thermodynamic processes
NEXT STEPS
- Study the first law of thermodynamics in detail
- Learn about isothermal processes and their characteristics
- Review calculations of work done in constant pressure and isothermal conditions
- Explore the relationship between pressure and volume in ideal gases
USEFUL FOR
Students studying thermodynamics, physics educators, and anyone interested in understanding the behavior of ideal gases in thermodynamic processes.