soul
- 61
- 0
Hi everyone,
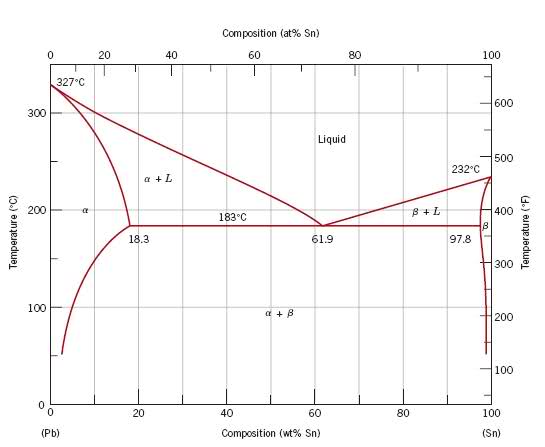
A lead–tin alloy of composition 30 wt% Sn–70 wt% Pb is slowly heated from a temperature
of 150 C (300F).
(a) At what temperature does the first liquid phase form?
(b) What is the composition of this liquid phase?
(c) At what temperature does complete melting of the alloy occur?
(d) What is the composition of the last solid remaining prior to complete melting?
It is probably an easy question, but I thought it is a good idea to consult first.
a) At 183 C, the first liquid phase forms.
b) We can draw a tie line and the point intersects with the liquidus line, gives us the composition of liquid. It is 61.9 wt % Sn.
c) It is around 250 C. Because phase diagram is on liquidus line.
d) Composition is 15wt % Sn. We can find it by drawing a tie line.
I am not sure about the question b and d. Because as far as I know composition of this liquid means that the composition of the components in this liquid. For instance, in some specific temperature liquid is composed of 30wt % Sn - 70 wt & Pb. However, in some sources I saw that they interpreted this statement as the composition of liquid phase in the total alloy. Like, again in some temperature, 30 % liquid- 70 % solid. Which one is correct. I am a bit confused. Any help will be greatly appreciated.
Thank you.
Homework Statement
A lead–tin alloy of composition 30 wt% Sn–70 wt% Pb is slowly heated from a temperature
of 150 C (300F).
(a) At what temperature does the first liquid phase form?
(b) What is the composition of this liquid phase?
(c) At what temperature does complete melting of the alloy occur?
(d) What is the composition of the last solid remaining prior to complete melting?
Homework Equations
The Attempt at a Solution
It is probably an easy question, but I thought it is a good idea to consult first.
a) At 183 C, the first liquid phase forms.
b) We can draw a tie line and the point intersects with the liquidus line, gives us the composition of liquid. It is 61.9 wt % Sn.
c) It is around 250 C. Because phase diagram is on liquidus line.
d) Composition is 15wt % Sn. We can find it by drawing a tie line.
I am not sure about the question b and d. Because as far as I know composition of this liquid means that the composition of the components in this liquid. For instance, in some specific temperature liquid is composed of 30wt % Sn - 70 wt & Pb. However, in some sources I saw that they interpreted this statement as the composition of liquid phase in the total alloy. Like, again in some temperature, 30 % liquid- 70 % solid. Which one is correct. I am a bit confused. Any help will be greatly appreciated.
Thank you.
Attachments
Last edited:
