Discussion Overview
The discussion revolves around understanding orbitals and energy levels within the context of the Bohr model of the atom. Participants explore the definitions and characteristics of different quantum numbers, the structure of orbitals, and the rules governing electron transitions between these energy levels.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
- Mathematical reasoning
Main Points Raised
- One participant recalls the relationship between principal quantum number (n) and the number of electrons in an energy level, noting that for n=1, there are 2 electrons.
- Another participant expresses uncertainty about how to determine the specific orbitals (s, p, d, etc.) associated with these energy levels.
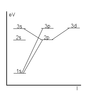
- A participant mentions using the Bohr model to analyze possible electron transitions and seeks clarification on how to determine the change in magnetic quantum number (Δm_l) during these transitions.
- Several participants discuss the rules for quantum numbers, including the relationships between n, l, and m, and how they define energy states and orbital orientations.
- One participant explains the maximum number of electrons in each subshell and the significance of Pauli's exclusion principle in determining electron configurations.
- A later reply questions the likelihood of specific transitions occurring, particularly between the 1s and 2p, and 1s and 3p states, suggesting that there may be restrictions on these transitions.
Areas of Agreement / Disagreement
Participants generally agree on the definitions and relationships between quantum numbers and orbitals, but there is uncertainty regarding the specifics of electron transitions and the conditions under which they occur. The discussion remains unresolved regarding the likelihood of certain transitions.
Contextual Notes
Participants express varying levels of familiarity with terminology and concepts, indicating potential gaps in understanding. There are also references to specific rules and principles that may depend on further context or clarification.