CAF123
Gold Member
- 2,918
- 87
Homework Statement
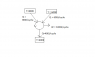
Explain whether or not the first or second law of thermodynamics are violated in the below cyclic process (see attachment)
Homework Equations
Clausius Inequality, Carnot efficiencies, ΔS ≥ 0 for a cyclic process.
The Attempt at a Solution
The first law is satisfied. The fact there are two hot reservoirs instead of one is putting me off a little bit since the K-P version of the second law only applies to heat entering the engine from a single hot reservoir. The generalized Clausius' inequality says that ##\sum_i \frac{\delta Q_i}{T_i} \leq 0 = \frac{600}{600} + \frac{800}{800} - \frac{400}{400} = 1## which is not less than zero. So the second law is violated. Is this all?
I was wondering if I could replace the two separate hot reservoirs with a single reservoir at 1400K? When I do this, I end up with ##\eta_C = \eta_E## which is not a violation of the second law, so I must have gone wrong in my attempts.
Many thanks.