Discussion Overview
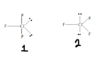
The discussion revolves around the molecular geometry of chlorine trifluoride (ClF3), specifically why it adopts a T-shaped structure rather than a trigonal planar configuration. The conversation touches on concepts from VSEPR theory and the spatial requirements of electron pairs.
Discussion Character
- Technical explanation, Conceptual clarification, Debate/contested
Main Points Raised
- One participant asserts that ClF3 has a trigonal bipyramidal arrangement and T-shaped molecular geometry, questioning why it could also be considered trigonal planar.
- Another participant explains that according to VSEPR theory, non-bonding electron pairs are sterically more demanding than bonding pairs, which influences the molecular shape.
- A participant seeks clarification on the term 'sterically more demanding than bound electron pairs' and expresses difficulty in understanding its meaning.
- In response, another participant states that non-bonding pairs need more space and occupy a larger volume compared to bonding pairs.
Areas of Agreement / Disagreement
Participants express differing views on the molecular geometry of ClF3, with no consensus reached on the applicability of the trigonal planar model.
Contextual Notes
The discussion highlights the complexity of molecular geometry and the influence of electron pair repulsion, but does not resolve the assumptions or definitions related to steric demands and VSEPR theory.