- #1
perplexabot
Gold Member
- 329
- 5
Hi all. Trying to find work done for a gas... Here is the question:
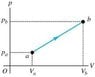
A quantity of air is taken from state a to state b along a path that is a straight line in the pv-diagram (figure is attached). If Va = 0.07, Vb = .11, Pa = 100,000 , Pb = 140,000. What is the work W done by the gas in this process? Assume that the gas may be treated as ideal.
Ok i know the area under the graph is work. But instead of doing it that way can i use this way?:
W = (integral of) p dV
and since p is changing linearly: p = mV + b where b = 0 and m = (Pb - Pa)/(Vb - Va) = k
so p = k* V
substituting that back into the Work equation i get:
W = (integral of) k * V dV
=> W = k * (integral of ) V dV
=> W = k/2 (Vb2 - Va2)
ok so after all this i end up with 3600 J which is wrong (actual answer is 4800). Can any1 tell me what I'm doing wrong? or why this technique doesn't work?
once again i know u can just find the area of the graph, but i want to know why this way is wrong..
Thanks all.
A quantity of air is taken from state a to state b along a path that is a straight line in the pv-diagram (figure is attached). If Va = 0.07, Vb = .11, Pa = 100,000 , Pb = 140,000. What is the work W done by the gas in this process? Assume that the gas may be treated as ideal.
Ok i know the area under the graph is work. But instead of doing it that way can i use this way?:
W = (integral of) p dV
and since p is changing linearly: p = mV + b where b = 0 and m = (Pb - Pa)/(Vb - Va) = k
so p = k* V
substituting that back into the Work equation i get:
W = (integral of) k * V dV
=> W = k * (integral of ) V dV
=> W = k/2 (Vb2 - Va2)
ok so after all this i end up with 3600 J which is wrong (actual answer is 4800). Can any1 tell me what I'm doing wrong? or why this technique doesn't work?
once again i know u can just find the area of the graph, but i want to know why this way is wrong..
Thanks all.
Attachments
Last edited: