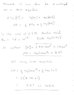- #1
gkangelexa
- 81
- 1
My book says that you cannot use the Henderson-Hasselbalch equation to find the pH at the equivalence point when titrating a weak acid with a strong base...
I was wondering why not?
it says that instead you must use the Kb of the conjugate base and that we find that from the Kw and the Ka.
Why?
and how do we find the Ka?
I was wondering why not?
it says that instead you must use the Kb of the conjugate base and that we find that from the Kw and the Ka.
Why?
and how do we find the Ka?