- #1
Orthoceras
- 117
- 43
- TL;DR Summary
- What does the energy diagram of a coated cathode with tunneling look like?
I am wondering about an exercise exam question (it isn't homework): "at low temperatures (<2000 K), thermionic emission of a tungsten cathode depends on tunneling. By coating the tungsten with a suitable substance, the emission by tunneling can be greatly increased. Question: which two properties of the coating maximise the tunneling? Answer: the coating should be thin compared to the wavelength of the electrons, and the work function of the coating should be less than the work function of tungsten."
This exercise doesn't require the student to explain this further. However, I am curious about the shape of the barrier in an energy diagram. Below see my attempt in which the electron is tunneling through a vacuum barrier towards a positive anode. This attempt isn't very satisfactory. The exam answer seems to suggest the coating itself requires tunneling, so the coating seems to be an insulator, whereas in my diagram the coating is a conductor with a reduced work function. What should the energy diagram really look like?
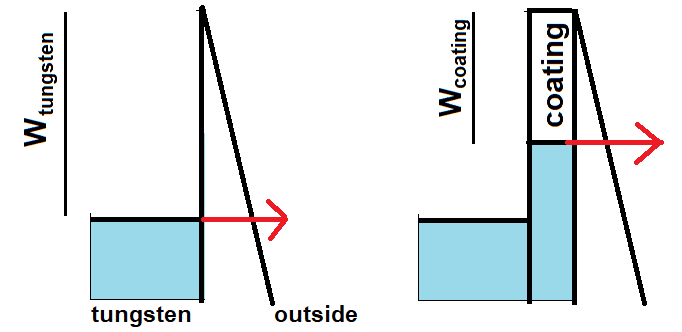
This exercise doesn't require the student to explain this further. However, I am curious about the shape of the barrier in an energy diagram. Below see my attempt in which the electron is tunneling through a vacuum barrier towards a positive anode. This attempt isn't very satisfactory. The exam answer seems to suggest the coating itself requires tunneling, so the coating seems to be an insulator, whereas in my diagram the coating is a conductor with a reduced work function. What should the energy diagram really look like?