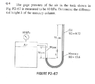
Calculate Pressure from Height & Specific Gravity
Click For Summary
Homework Help Overview
The discussion revolves around calculating pressure from height and specific gravity, focusing on the relationship between density, specific gravity, and pressure in fluid mechanics.
Discussion Character
- Exploratory, Conceptual clarification, Mathematical reasoning, Assumption checking
Approaches and Questions Raised
- Participants explore how to convert specific gravity into density and its application in pressure calculations. There are attempts to set up equations involving atmospheric pressure and gauge pressure, with questions about assumptions regarding the mass of air and the interpretation of pressure differences.
Discussion Status
The discussion includes various attempts to solve the problem, with some participants providing guidance on the definitions of gauge pressure and the relationship between pressure and height. There are multiple interpretations of the equations being used, and participants are actively questioning their calculations and assumptions.
Contextual Notes
Participants mention specific values for density and gauge pressure, and there is uncertainty regarding the assumptions made about atmospheric pressure and the mass of air in the calculations.
Similar threads
- · Replies 10 ·
- · Replies 7 ·
- · Replies 13 ·
- · Replies 4 ·
- · Replies 6 ·