SUMMARY
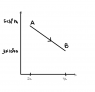
The discussion centers on calculating the work done by a gas during expansion on a pV diagram, specifically from point A to point B. The correct formula for work is identified as Work = PΔV. A participant calculated the area under the curve to be 8 x 10^11 J, while the answer key states the correct value is 800 J. The discrepancy arises from a misunderstanding of unit conversion, specifically mistaking liters for cubic meters.
PREREQUISITES
- Understanding of pV diagrams in thermodynamics
- Familiarity with the concept of work in physics
- Knowledge of unit conversions, particularly between liters and cubic meters
- Basic proficiency in calculus for area calculations
NEXT STEPS
- Study the principles of thermodynamics, focusing on gas laws
- Learn about unit conversions, especially between metric units
- Explore the calculation of areas under curves in physics
- Review examples of work done in various thermodynamic processes
USEFUL FOR
Students in physics, particularly those studying thermodynamics, as well as educators looking for clarification on work calculations in gas expansion scenarios.