ifihadsomebacon
- 4
- 0
Going from:
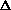 E = q + w
E = q + w
To:
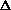 H =
H =
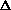 E +
E +
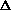 (PV)
(PV)
I'm confused as to why you add the product of the pressure and volume of the system to the internal energy to get enthalpy. Is it just because "that's what enthalpy is defined as"? I think I understand that when holding pressure constant, the
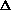 (PV) becomes P
(PV) becomes P
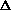 V and cancels with the w = P
V and cancels with the w = P
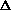 V from
V from
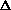 E, giving
E, giving
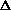 H = q. If pressure is not constant, does that mean
H = q. If pressure is not constant, does that mean
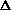 H = q + w +
H = q + w +
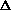 (PV) or
(PV) or
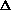 H = q + P
H = q + P
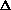 V +
V +
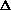 (PV) is true? What does this mean, practically? I just am wondering why are there two "PV" expressions in the first place? What even is
(PV) is true? What does this mean, practically? I just am wondering why are there two "PV" expressions in the first place? What even is
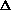 (PV)? It doesn't make sense to me like P
(PV)? It doesn't make sense to me like P
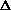 V, what about
V, what about
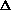 (PV) makes it so it can be added on initially?
(PV) makes it so it can be added on initially?
I know I asked a lot of questions, I'm just trying to make my confusion clear. (Ha)
To:
I'm confused as to why you add the product of the pressure and volume of the system to the internal energy to get enthalpy. Is it just because "that's what enthalpy is defined as"? I think I understand that when holding pressure constant, the
I know I asked a lot of questions, I'm just trying to make my confusion clear. (Ha)
Attachments
-
 delta.gif63 bytes · Views: 2,985
delta.gif63 bytes · Views: 2,985 -
 delta.gif63 bytes · Views: 3,144
delta.gif63 bytes · Views: 3,144 -
 delta.gif63 bytes · Views: 2,962
delta.gif63 bytes · Views: 2,962 -
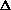 delta.gif63 bytes · Views: 3,056
delta.gif63 bytes · Views: 3,056 -
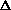 delta.gif63 bytes · Views: 2,898
delta.gif63 bytes · Views: 2,898 -
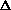 delta.gif63 bytes · Views: 2,764
delta.gif63 bytes · Views: 2,764 -
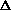 delta.gif63 bytes · Views: 2,661
delta.gif63 bytes · Views: 2,661 -
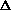 delta.gif63 bytes · Views: 2,748
delta.gif63 bytes · Views: 2,748 -
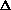 delta.gif63 bytes · Views: 2,766
delta.gif63 bytes · Views: 2,766 -
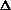 delta.gif63 bytes · Views: 2,753
delta.gif63 bytes · Views: 2,753 -
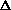 delta.gif63 bytes · Views: 2,787
delta.gif63 bytes · Views: 2,787 -
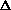 delta.gif63 bytes · Views: 2,622
delta.gif63 bytes · Views: 2,622 -
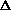 delta.gif63 bytes · Views: 2,782
delta.gif63 bytes · Views: 2,782 -
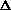 delta.gif63 bytes · Views: 2,748
delta.gif63 bytes · Views: 2,748 -
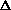 delta.gif63 bytes · Views: 2,646
delta.gif63 bytes · Views: 2,646 -
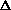 delta.gif63 bytes · Views: 2,767
delta.gif63 bytes · Views: 2,767 -
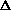 delta.gif63 bytes · Views: 2,800
delta.gif63 bytes · Views: 2,800