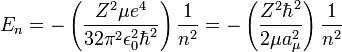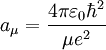Discussion Overview
The discussion revolves around the energy levels of helium and helium-like atoms, particularly in relation to the energy levels of hydrogen-like atoms. Participants explore the complexity of deriving equations that fit the observed spectrum of helium and the methods used to obtain these energy levels.
Discussion Character
- Exploratory, Technical explanation, Debate/contested
Main Points Raised
- One participant presents the equation for energy levels of hydrogen-like atoms and questions the corresponding equation for helium or helium-like atoms, suggesting that helium's spectrum may resemble two hydrogen spectra superimposed.
- Another participant asserts that exact solutions for helium-like systems are not possible, indicating that only two-body systems can be solved exactly.
- A participant clarifies that they are not seeking a solution but rather an equation that fits the empirically observed spectrum.
- There is a contention regarding the definition of a solution, with one participant suggesting that fitting an experimental spectrum might involve complex mathematical constructs, which could be impractical.
- Another participant mentions that in practice, researchers typically refer to tables or charts for energy levels or utilize computer simulations, referencing the work of Gordon W. F. Drake for accurate calculations of helium.
- It is noted that understanding Drake's calculations requires advanced knowledge of quantum mechanics.
Areas of Agreement / Disagreement
Participants express differing views on the feasibility of deriving an equation for helium's energy levels, with some suggesting that empirical fitting methods may not be practical. The discussion remains unresolved regarding the specific equations or models that could be used.
Contextual Notes
Limitations include the complexity of helium's energy levels compared to hydrogen-like atoms and the potential impracticality of fitting experimental spectra with simple equations.