Slimjimi
- 5
- 0
I am trying to determine whether the evaporation rate of a 3L erlenmeyer flask would be negligible given the following circumstances. The flask would have a rubber stopper in it, with a hole approximately 1mm in radius greater than the steel drip line feeding into it (leaving an open ring that has an outer diameter of ~8mm and an inner diameter of ~6mm). I would like to take realtime mass measurements as the drip line does it's thing. The process would span several days, so evaporation has to be taken into account. The process is drawn roughly here:
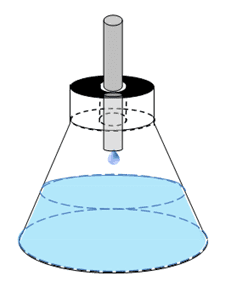
Thanks a lot!
Thanks a lot!