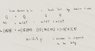gibson101
- 47
- 0
My answer is 12.9 grams, and the correct answer is supposed to be 11.9 grams. I cannot figure out why? Note. the answer i got is 12.9, and not 11.06. I know for this situation, the heat absorbed by the ice equals the heat lost by the water and aluminum, and since there is a phase change from a solid to liquid then i need the heat of fusion constant for solid water, which in kcal/kg which is 79.7. Now I am thinking that this is where my mistake is, because temperature remains constant during a phase change. Therefore, I cannot put 16-(-8.7)? I have to write it out into two separate m*c*deltaT's?
Attachments
Last edited: