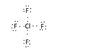Discussion Overview
The discussion revolves around drawing the Lewis Dot Structure for the ClF4+ ion, focusing on the challenges of accommodating additional electrons while adhering to or deviating from the octet rule. Participants explore the implications of using higher-energy orbitals and the role of atomic period in determining electron distribution.
Discussion Character
- Homework-related
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
- One participant calculated a total of 34 electrons for the Lewis structure of ClF4+, considering the contributions from chlorine and fluorine atoms and the positive charge.
- Another participant suggested that the lone pair should be placed on the chlorine atom, indicating that the octet rule may not apply due to the involvement of higher-energy orbitals.
- There is a discussion about whether chlorine, being in the third period, can utilize d orbitals to accommodate extra electrons, with one participant affirming this possibility.
- One participant questioned if there are general rules for distributing extra electrons to atoms capable of supporting d orbitals, particularly for elements in period 3 or higher.
- A later reply cautioned against the existence of "hard & fast rules" for Lewis structures, emphasizing that they are simplifications and that quantum chemistry provides a deeper understanding of molecular structures.
- Symmetry was mentioned as a potential guide for determining electron distribution, with concerns raised about the implications of allowing fluorine atoms to gain extra electrons leading to complex resonance structures.
Areas of Agreement / Disagreement
Participants express differing views on the application of the octet rule and the use of d orbitals in Lewis structures. There is no consensus on a definitive rule for handling extra electrons in such scenarios.
Contextual Notes
Limitations include the simplifications inherent in Lewis diagrams and the need for a more nuanced understanding through quantum chemistry to fully grasp molecular structures.