Klacid
- 2
- 0
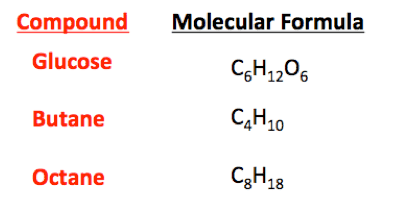
Hey guys i wanted to know how make some chemicals when i have an equation, for example:
The discussion revolves around the process of creating chemicals based on chemical equations and molecular formulas. Participants explore the relationship between molecular formulas and chemical reactions, as well as the prerequisites for synthesizing compounds.
Participants do not reach a consensus on the best methods for synthesizing chemicals from equations, and multiple viewpoints regarding the interpretation of molecular formulas and their implications for chemical reactions remain present.
Participants acknowledge that the discussion involves complexities such as the nature of isomers, the necessity of specific precursors, and the conditions under which reactions may or may not occur.
First of all, you don't have any equations in the graphic above. These are called the molecular formulas for the various compounds shown.Klacid said:Hey guys i wanted to know how make some chemicals when i have an equation, for example: