Discussion Overview
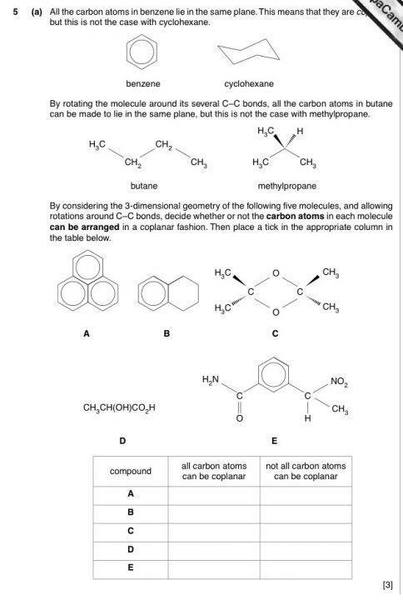
The discussion revolves around the coplanarity of carbon atoms in various molecular structures, specifically focusing on the arrangements in compounds labeled B, C, D, and E, as well as methylpropane. Participants explore the implications of hybridization (sp3 and sp2) and the geometric arrangements of carbon atoms.
Discussion Character
- Homework-related
- Exploratory
- Debate/contested
- Technical explanation
Main Points Raised
- Some participants assert that in compound E, carbon atoms are not coplanar, with one being able to move freely, potentially aligning with others at times.
- There is a question regarding the number of carbon atoms in compound D and whether they can be arranged in a way that they do not lie on a single surface.
- Participants discuss that while carbon atoms in benzene are coplanar due to sp2 hybridization, the sp3 hybridized carbons in cyclohexane are tetrahedral, raising questions about their coplanarity.
- One participant suggests that the central COCO ring in compound C might be flat, but expresses uncertainty about the arrangement of carbon atoms.
- Another participant states that any three points are coplanar, but raises confusion about why the carbon atoms in methylpropane cannot all be coplanar.
- It is mentioned that the carbon atoms in methylpropane cannot all lie in the same plane due to the tetrahedral geometry of sp3 hybridized carbons, with one carbon atom needing to stick out.
- Participants suggest using physical models to better visualize the molecular structures and their arrangements.
Areas of Agreement / Disagreement
Participants express uncertainty and confusion regarding the coplanarity of carbon atoms in various compounds, particularly in methylpropane. There is no consensus on the specific arrangements or the implications of hybridization on coplanarity.
Contextual Notes
Participants reference various molecular structures and hybridization types, but the discussion lacks definitive conclusions about the coplanarity of carbon atoms in the mentioned compounds. The reasoning relies on geometric and hybridization principles that may not be fully resolved.