Homework Help Overview
The discussion revolves around a multiple-choice question related to heat transfer and specific heat capacity, involving two identical heaters and their effects on different masses of substances.
Discussion Character
- Exploratory, Conceptual clarification, Assumption checking
Approaches and Questions Raised
- Participants discuss the relationship between heat absorbed and the properties of the substances involved. Questions arise regarding the equality of heat transfer (Q1 = Q2) and the implications of mass differences on calculations.
Discussion Status
Some participants have provided hints and guidance on relevant formulas and concepts, while others are exploring different interpretations of the problem. There is an ongoing examination of assumptions regarding mass and energy transfer.
Contextual Notes
Participants are reminded to show their work, and there is a focus on the specific heat capacity and the relationship between power and heat transfer. The original poster acknowledges a misunderstanding of the question's requirements.
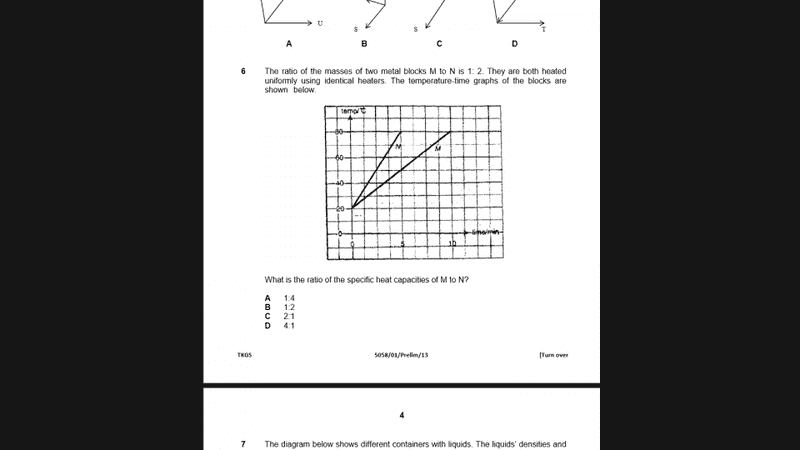 Hello! I need help with this question. The answer is D but I don't understand why :/ Thanks!
Hello! I need help with this question. The answer is D but I don't understand why :/ Thanks!