Pliskin01
- 1
- 0
Hello all, this is my first post here and I am glad to be a new member of the PhysicsForums community! I have a question which I believe will be a good challenge for the bright minds here:
I am trying to create a system of Al-Air batteries to power a motor which will be hooked up to an rc car chassis and carry a sizable load. I've constructed the system seen in the following picture:
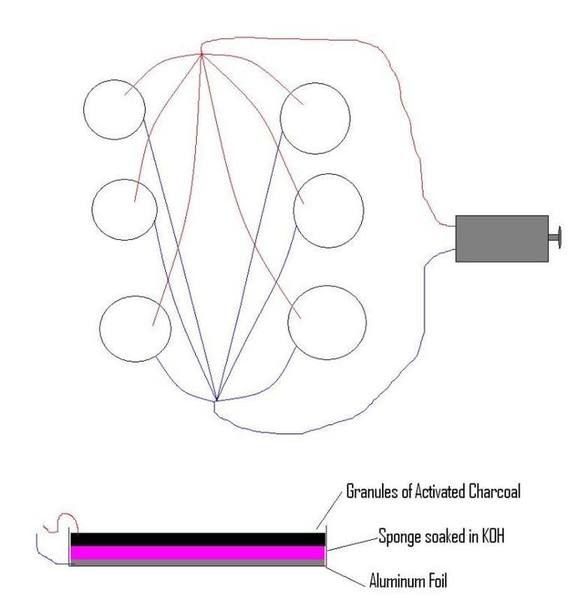

each circle represents 3 Al-Air battery cells in a stack connected to each other in series. These stacks are then connected to the other 5 stacks in parallel. Using this set-up, I am able to get readings of 1.6A and 3.0V. While this is enough to turn the motor, it is not enough for the motor to pull any reasonable load placed on it.
The motor requirements are 750mA and 7.5V
Since the current requirement is being met, the voltage just needs to be increased to make the motor turn faster/more strongly (is this correct?).
Is there any way to increase the voltage or make a more efficient battery setup? I am already using 18 cells (6 stacks of 3).
Thank you for reading, any help is appreciated!
I am trying to create a system of Al-Air batteries to power a motor which will be hooked up to an rc car chassis and carry a sizable load. I've constructed the system seen in the following picture:
each circle represents 3 Al-Air battery cells in a stack connected to each other in series. These stacks are then connected to the other 5 stacks in parallel. Using this set-up, I am able to get readings of 1.6A and 3.0V. While this is enough to turn the motor, it is not enough for the motor to pull any reasonable load placed on it.
The motor requirements are 750mA and 7.5V
Since the current requirement is being met, the voltage just needs to be increased to make the motor turn faster/more strongly (is this correct?).
Is there any way to increase the voltage or make a more efficient battery setup? I am already using 18 cells (6 stacks of 3).
Thank you for reading, any help is appreciated!
Last edited: